If you have ever been curious about ‘does water conduct electricity?’? )You are not alone. This is one of the most common questions in science classrooms and daily life – but the answer is often confusing: some say water is a conductor, while others say pure water is an insulator.
Actually, both statements are correct. The key is to distinguish between “pure water” and “water containing impurities”. This guide will unravel the mystery for you, not only explaining the scientific principles of water conductivity, but also showing you how to measure the conductivity of water, why it is important, and what the conductivity of water means in different scenarios.

Does Water Conduct Electricity? —Short Answer
The answer is: both conductive and non-conductive – it all depends on what is in the water.
If you are holding absolutely pure water – such as ultrapure water used in semiconductor manufacturing or pharmaceutical production – it is actually an excellent insulator with conductivity close to zero.
But the water you drink, use for bathing, and in the fish tank is not pure. They contain dissolved minerals, salts, and other impurities. It is these dissolved substances that make water conductive.
Key points:
Water molecules (H ₂ O) are neutral and do not possess a charge. Ions, which are charged particles that have the potential to move, are necessary for the transmission of electric current through water.
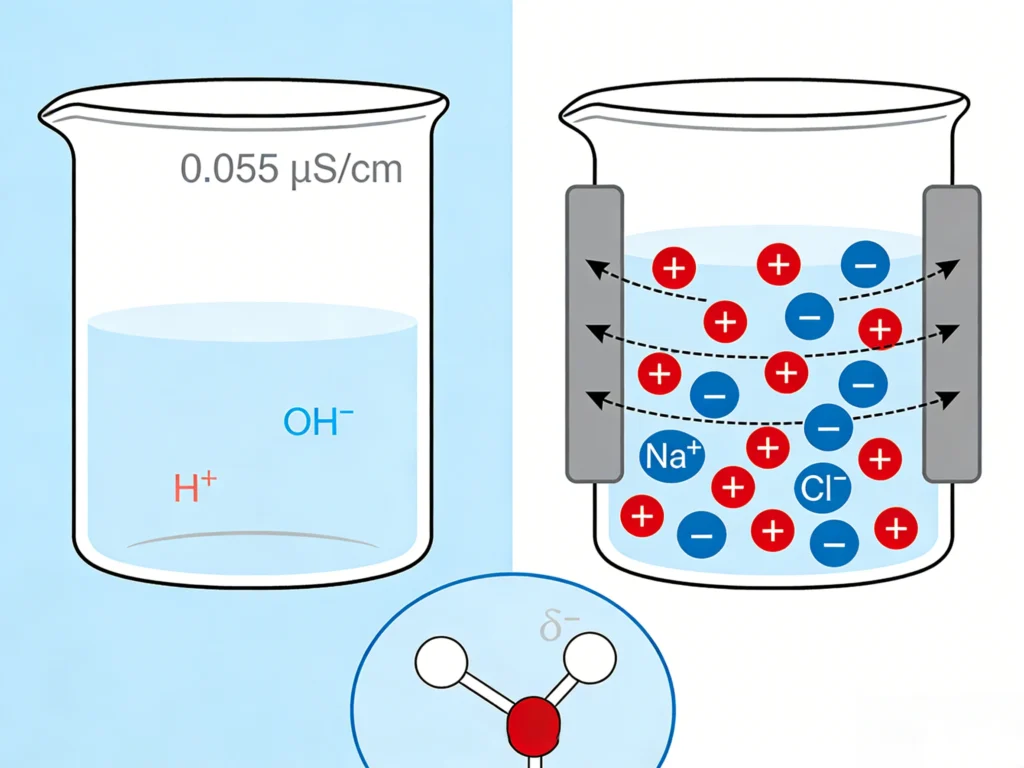
Salt (sodium chloride) dissolves in water, forming positively charged Na ions (Na ⁺) and negatively charged chloride ions (Cl ⁻). These small ‘charge transporters’ move toward the opposing electrode when a voltage is applied, creating an electric current.
The reason:
- No ions in pure water = non-conductive.
- High ion content in salt water results in high conductivity.
- Sugar water is nonconductive due to the absence of ions.
Electricity is not conducted by water; rather, it is conducted by dissolved substances.

Classic Experiment – Salt vs. Sugar
Let’s first conduct a simple experiment that can reveal all the facts you need to know.
Need to prepare:
- Two glass beakers
- Pure water (distilled water or deionized water)
- Salt (sodium chloride)
- white granulated sugar
- Conductivity meter or multimeter
Steps for the experiment:
Both beakers are full of clean water. Conductivity measurement: The conductivity meter shows a very high resistance (over 900,000 ohms). Water that is pure does not conduct electricity.
Put salt in the first beaker and mix it up. Retest: The resistance goes down a lot (below 80000 ohms). Salt water is a good conductor.
Put sugar in the second beaker and mix it up. Retest: the resistance is still high. Sugar water does not carry electricity.
What happened?
Salt (sodium chloride) dissolves in water and decomposes into ions – positively charged sodium ions (Na ⁺) and negatively charged chloride ions (Cl ⁻). These charged particles can move freely in water and carry an electric current.
Sugar is formed by covalent bonding of carbon, hydrogen, and oxygen atoms. Atoms share electrons and do not produce ions. Sugar exists in neutral molecular form after dissolution – without a charge carrier, there is no conductivity.
Key conclusion: The conductivity of water comes from ions, not water molecules themselves.

Scientific Principles – Why Pure Water is an Insulator
The structure of water molecules
Water molecules (H ₂ O) are polar molecules – with one end carrying a slight positive charge and the other end carrying a slight negative charge. This polarity makes water an excellent solvent, but does not make it conductive.
To make current flow, there must be movable charged particles. In pure water, the only charged particles are H ⁺ and OH ⁻ ions produced by the weak ionization of water itself. At 25 ° C, the concentration of these two ions in pure water is only 10 ⁻⁷ mol/L – an extremely low value.
That’s why the theoretical conductivity of chemically pure water is only 0.055 µ S/cm – in fact, it’s an insulator.
Electronic conductivity vs. Ionic conductivity
In metals, current is transmitted through the movement of electrons. Copper wire can conduct electricity because electrons can move freely within the metal lattice.
In water, electrons cannot move freely. Electric current is transmitted through the movement of ions – atoms or molecules that gain or lose electrons and become charged.
This is called ion conduction, which is fundamentally different from electronic conduction in metals.
The role of ions
When a voltage is applied between two electrodes immersed in water, positive ions (cations) are attracted to the negative electrode, and negative ions (anions) are attracted to the positive electrode. Their movement creates an electric current.
The more ions and the stronger the migration ability, the higher the conductivity.
What makes water conductive?
Source of ions in water
Most natural water and tap water contain ionizable dissolved substances:
| Source | Common Ions | Typical Conductivity Range |
|---|---|---|
| Dissolved minerals | Ca²⁺, Mg²⁺, Na⁺, K⁺, HCO₃⁻, SO₄²⁻, Cl⁻ | 50–1500 µS/cm |
| Dissolved salts | Na⁺, Cl⁻ | Highly variable, can exceed 50,000 µS/cm |
| Industrial discharge | Various ions (heavy metals, acids, bases) | Highly variable, can be extremely high |
| Agricultural runoff | Nitrates (NO₃⁻), Phosphates (PO₄³⁻), K⁺ | 250–3000 µS/cm |
| Sewage/wastewater | NH₄⁺, Cl⁻, PO₄³⁻, organic ions | 500–3000 µS/cm |
| Atmospheric deposition | H⁺, SO₄²⁻, NO₃⁻ (acid rain) | 5–100 µS/cm |
| Seawater intrusion | Na⁺, Cl⁻, Mg²⁺, SO₄²⁻ | >3000 µS/cm |
Important things that affect conductivity
The amount of ions
In weak solutions, the amount of ions is directly related to the amount of conductivity. The conductivity goes up as the number of ions goes up.
Type of ion
various ions have various levels of conductivity. Ions that are small and move quickly, like H+ and OH-, conduct electricity considerably better than ions that are big and move slowly.
The temperature
The conductivity goes up by about 2% for every 1 °C rise in temperature. When water is heated, ions flow faster, which makes the current transmission more effective.
How thick the solution is
A solution with a higher viscosity will make ions flow more slowly and lower conductivity.
Why Conductivity is Important – Practical Applications
Conductivity is the ‘controlling hand’ of various industries:
| Sector | One-Line Summary |
|---|---|
| Water Quality Monitoring | Conductivity change = contamination warning |
| Industrial Control | Prevents scaling, corrosion, protects chips, ensures compliance |
| Agriculture | Nutrient solution accuracy? Conductivity decides |
| Aquaculture | Conductivity spike = fish are screaming for help |
| Desalination | Membrane failed? Rejection rate? One test tells all |
Conductivity lets you detect problems before they occur, and maintain control before you lose it.
👉 Sino-Inst Conductivity Measurement Solutions

Conductivity of Different Waters – Reference Comparison Table
| Water Type | Typical Conductivity (µS/cm) | Notes |
|---|---|---|
| Ultrapure water | 0.055 | Theoretical pure water — excellent insulator |
| Distilled water | 0.5–3 | Good insulator, but absorbs CO₂ from air, forming carbonic acid and increasing conductivity slightly |
| Deionized water | 0.1–10 | Varies based on purification quality |
| Rainwater | 5–30 | Naturally soft, low mineral content |
| Meltwater (glaciers/snow) | 20–50 | Very low mineral content |
| Reverse osmosis (RO) water | 10–100 | Depends on membrane efficiency |
| Drinking water (tap) | 50–1500 | Highly variable depending on source and treatment |
| Freshwater rivers/lakes | 100–2000 | Depends on geology of watershed |
| Agricultural irrigation water | 250–3000 | Higher conductivity may indicate salinity issues |
| Groundwater | 500–5000 | Mineral content varies by aquifer |
| Brackish water | 3000–50,000 | Mix of fresh and salt water |
| Seawater | ~50,000 | Approximately 50 mS/cm — excellent conductor |
| Industrial wastewater | Highly variable | Can be extremely high (10,000–100,000+) |
FAQ
Related Products
Conclusion: From “Can water conduct electricity” to “Controllable water quality”
Water itself is non-conductive, only ions dissolved in water are conductive. Conductivity is the ruler for measuring these ions – it allows you to warn before pollution, maintain before damage, and adjust before death.
Sino Inst transforms’ invisible water quality ‘into’ visible data ‘. Focusing on water quality measurement for 20 years, from portable to online, there is always one that suits you.
👉 View Sino Inst Conductivity Meter
Don’t guess, just test it and you’ll know.
Request A Quote
More Resources
-
Top 10 Portable Carbon Monoxide Detector for Ice Fishing
Authored by Sino-Inst. We are a leading manufacturer and supplier of professional gas detection, flow measurement, and industrial instrumentation solutions. With decades of…
-
What Should the Salt Level Be in My Pool?(2026 Guide)
Having a swimming pool should be a pure enjoyment, but it can also bring many troubles. Many pool owners may ask: What should…
-
Top 7 Carbon Monoxide Detector Manufacturers USA 2026
Authored by Sino-Inst. We are a professional supplier of industrial process and analytical instruments, dedicated to providing reliable measurement and analysis solutions while…
-
6 Different Types of Gas Monitors for Industrial Safety
Authored by the engineering team at Sino-Inst. As a professional supplier of industrial process and analytical instruments, we specialize in helping global clients…
-
6 Best Furnace Combustion Analyzer Models for 2026: Expert Guide
Achieving optimal thermal efficiency and ensuring strict safety compliance in heating systems requires precise diagnostic equipment. Whether you are servicing residential HVAC units…
-
How to Lower the pH in a Pool Safely? (2026)
Learning how to lower the pH in a pool is the first step in ensuring clear water quality, whether in a private backyard…
.png)


















